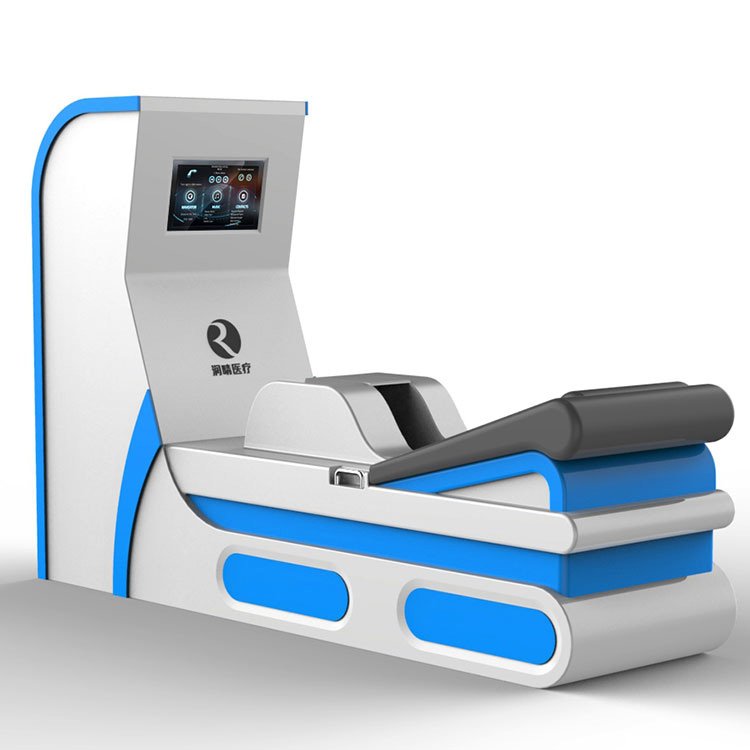
Selecting the right colon hydrotherapy equipment represents a significant investment for UK healthcare facilities. I have spent considerable time researching the Dotolo brand to provide you with actionable intelligence for your procurement decisions. This analysis examines Dotolo’s market position, technical specifications, regulatory compliance, and practical alternatives available to UK buyers in 2024.
The colon hydrotherapy equipment market presents numerous options. Your choice impacts patient outcomes, operational costs, and long-term business viability. Understanding what Dotolo offers—and where alternatives might better serve your specific requirements—empowers informed decision-making.
Throughout this evaluation, I reference comparative data from established brands including Libbe colonic machines, Hydrosan systems, and emerging manufacturers serving the UK market. Each section addresses questions UK practitioners consistently raise when evaluating Dotolo colon hydrotherapy machines.
Dotolo Brand Heritage and Market Position
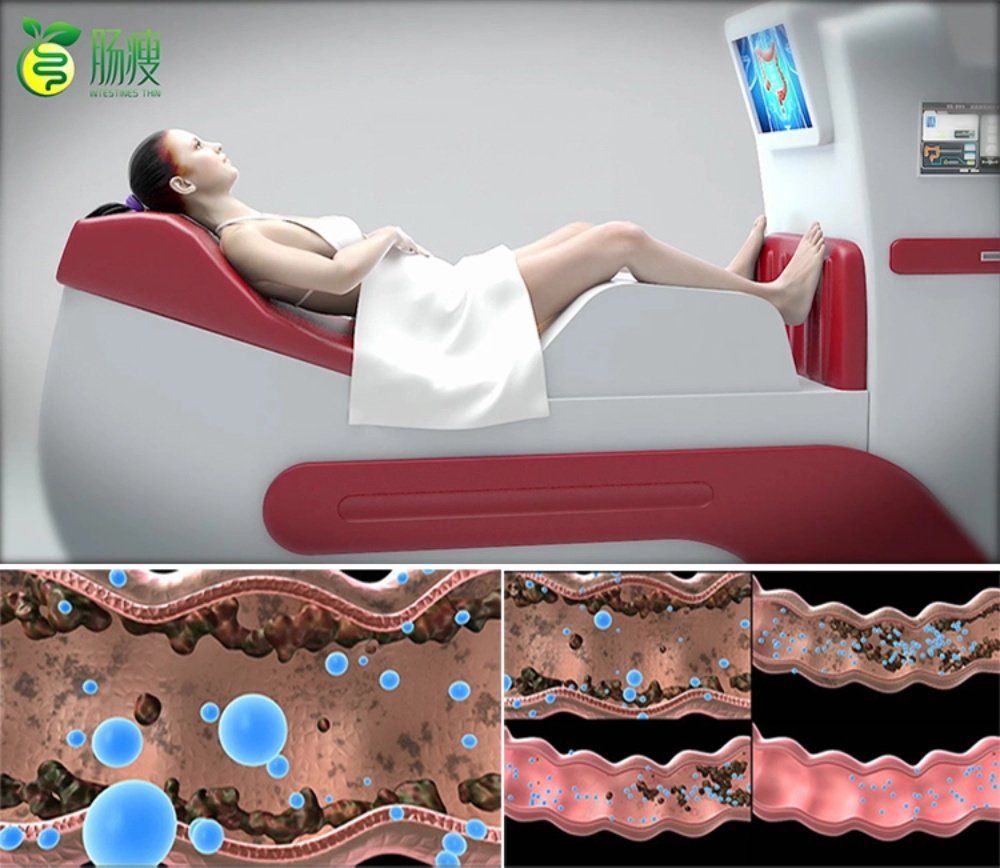
Dotolo Research Corporation established its reputation manufacturing colon hydrotherapy devices primarily for the North American market. The company built recognition through its Toxygen model, which became synonymous with closed system colonic machines in wellness and medical facilities.
European distribution operates through Dotolo Europe Ltd., based in Prague. This arrangement creates specific considerations for UK buyers navigating post-Brexit import procedures. The brand positions itself in the premium segment, competing with established European manufacturers.
Dotolo’s Market Strengths
- Established brand recognition in wellness sector
- Long operational history demonstrating business stability
- CE certification for European market compliance
- Integrated ozone therapy capabilities in select models
- Comprehensive training programmes for practitioners
Considerations for UK Buyers
- Premium pricing structure above market average
- European distributor adds intermediary layer
- Limited UK-based technical support infrastructure
- Longer lead times for delivery and servicing
- Spare parts procurement through continental Europe
The Dotolo colon hydrotherapy machine range includes the Toxygen model and the Hydromat Comfort system. Both represent closed system approaches, offering practitioner control over water temperature, pressure, and flow rate. This design philosophy appeals to clinics prioritising therapist-guided sessions over open-system alternatives.
Understanding where Dotolo fits within the broader colonic machine market helps contextualise your investment. The brand commands respect but operates in an increasingly competitive landscape with emerging manufacturers offering comparable technology at varied price points.
Technical Specifications and Equipment Features Analysis
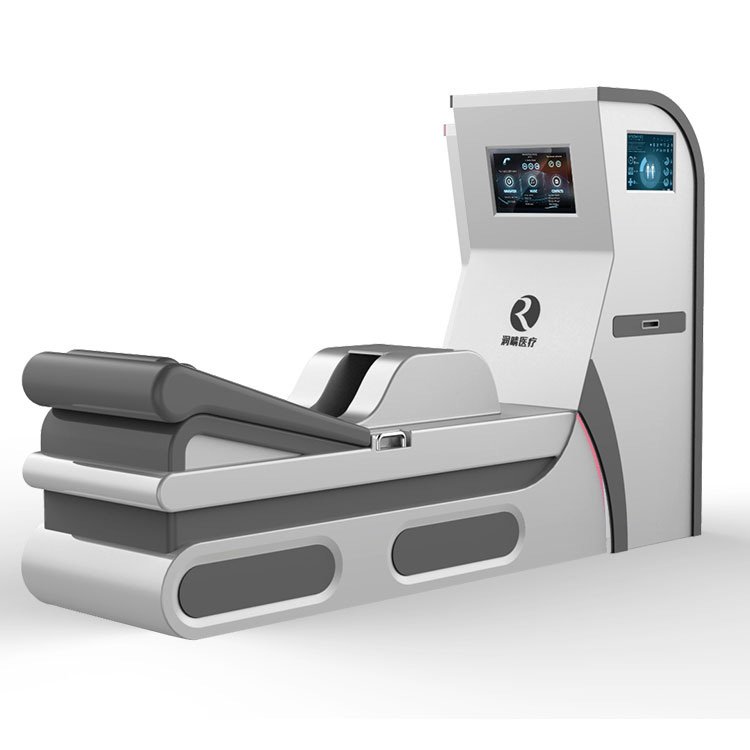
Evaluating colon hydrotherapy machines requires examining specific technical parameters that influence treatment efficacy and operational reliability. I have compiled comparative data to illustrate how Dotolo equipment specifications align with industry standards.
Dotolo Hydromat Comfort Core Specifications
| Parameter | Specification | Clinical Significance |
| Water Temperature Range | 38°C – 41°C | Maintains physiological comfort zone |
| Water Pressure Control | 0 – 0.6 Pa variable | Adjustable for patient sensitivity |
| Filtration System | Multi-stage activated carbon | Removes chlorine and contaminants |
| Session Duration Capacity | 30-45 minutes typical | Standard therapeutic window |
| Power Requirements | 220-240V UK standard | Direct compatibility with UK electrical systems |
| Footprint Dimensions | Approximately 2.5m x 1.7m | Requires dedicated treatment room space |
The Hydromat Comfort incorporates a touchscreen control interface, simplifying operation for practitioners. Water heating occurs automatically, with safety cutoffs preventing temperature excursions. The disposable speculum system addresses infection control requirements critical in UK healthcare settings.
Regulatory Compliance Note: All Dotolo equipment marketed in the UK must display CE marking indicating conformity with European Medical Device Regulation (EU MDR 2017/745). Verify certification documentation before procurement to ensure regulatory compliance for your facility.
Operational Features Comparison
Comparing operational capabilities helps identify which features deliver practical value versus marketing differentiation. I have assessed features against typical clinic requirements:
- Waste Viewing System: Dotolo’s transparent waste tube allows visual monitoring. Some practitioners value this; others find open system designs more patient-friendly.
- Odour Management: Integrated carbon filtration addresses treatment room air quality. Essential feature for facilities with multiple treatment rooms.
- Water Flow Control: Manual adjustment versus automated peristaltic pumps represents a design choice. Dotolo uses therapist-controlled flow rates.
- Ozone Integration: Select models incorporate medical ozone therapy capabilities, adding functional versatility for progressive practitioners.
- Sanitation Protocols: Automated tube cleaning cycles reduce cross-contamination risk and simplify hygiene procedures.
Understanding how colonic machines work at a technical level helps you evaluate whether Dotolo’s specific implementation aligns with your therapeutic approach and patient demographic.
Pricing Structure and Total Cost of Ownership
Investment analysis extends beyond initial equipment purchase. I have examined the complete financial picture UK buyers face when considering Dotolo colonic machines.
Initial Purchase Investment
Dotolo equipment typically positions in the premium price bracket. While exact pricing fluctuates based on configuration and distributor negotiations, UK buyers generally encounter:
- Base Equipment Cost: £12,000 – £18,000 for Hydromat Comfort depending on specifications
- Shipping and Import: £800 – £1,500 from European distributor to UK location
- Installation Services: £500 – £1,200 for professional setup and commissioning
- Initial Training: £600 – £1,000 per practitioner for certification programmes
- First-Year Consumables: £400 – £800 for disposable speculums, tubing, filters
Total First-Year Investment: Expect £14,300 – £22,500 all-inclusive for a fully operational Dotolo system in your UK facility. This positions Dotolo in the upper quartile of market pricing.
For perspective on market pricing ranges, reviewing colonic machine price comparisons across brands provides valuable context for your budgeting process.
Ongoing Operational Costs
Beyond initial outlay, recurring expenses impact long-term profitability:
| Cost Category | Annual Estimate (GBP) | Notes |
| Consumable Supplies | £1,200 – £2,000 | Based on 500-800 treatments annually |
| Maintenance Service | £400 – £800 | Annual inspection and calibration |
| Replacement Filters | £150 – £300 | Water filtration system components |
| Electricity Consumption | £180 – £250 | Based on average UK commercial rates |
| Insurance Premium Addition | £200 – £400 | Equipment value coverage addition |
| Extended Warranty (Optional) | £600 – £1,000 | Beyond standard 1-year coverage |
Five-year total cost of ownership for a Dotolo system typically ranges £20,000 – £32,000 depending on utilisation intensity and warranty decisions. This financial commitment requires careful evaluation against your projected treatment volume and revenue targets.
Seeking Cost-Effective CE-Certified Alternatives?
UK healthcare facilities increasingly explore equipment options delivering comparable functionality at more accessible price points. MAIKONG manufactures CE-certified colon hydrotherapy systems specifically designed for the UK and European markets, offering professional-grade features with transparent pricing and streamlined delivery.
Dotolo Alternatives: Comprehensive Brand Comparison for UK Buyers

No single manufacturer dominates the colon hydrotherapy equipment market. I have evaluated prominent alternatives UK buyers consider alongside Dotolo to provide comprehensive comparison data.
Established European and North American Brands
Libbe Colonic System
The Libbe colonic machine pioneered open-system design, offering patient privacy and self-administered sessions. UK practitioners serving wellness-focused clientele often prefer this approach.
- Open-system design with gravity-fed water flow
- Lower equipment costs than Dotolo (£8,000-£12,000)
- Simpler operation requiring less intensive training
- Patient preference for privacy and control
- Reduced practitioner time per session
Consideration: Open systems suit wellness applications but may not satisfy medical practitioners preferring controlled therapeutic protocols.
Hydrosan Colon Hydrotherapy Equipment
German-engineered Hydrosan systems compete directly with Dotolo in the professional healthcare segment, emphasising precision engineering and medical-grade construction.
- Closed-system design similar to Dotolo approach
- Comparable pricing (£13,000-£19,000 range)
- Strong European service network
- Advanced digital controls and data logging
- Established UK distributor relationships
Consideration: Hydrosan offers similar capabilities with potentially better UK support infrastructure than Dotolo’s Prague-based distribution.
Angel of Water Colonic Systems
Angel of Water equipment positions as a mid-market option, balancing professional features with more accessible pricing for independent practitioners and smaller facilities.
- Hybrid system design combining open and closed elements
- Mid-range pricing (£7,000-£11,000)
- Compact footprint suitable for smaller treatment rooms
- CE certification for UK market compliance
- Growing presence in UK wellness sector
Consideration: Suitable for budget-conscious buyers accepting trade-offs in advanced features and brand prestige.
Toxygen Professional Systems
Originally a Dotolo model name, Toxygen now represents a product category. Understanding this naming can prevent confusion during procurement research.
- Refers to Dotolo’s flagship closed-system model
- Integrated medical ozone therapy capability
- Professional-grade construction and components
- Premium pricing reflecting advanced features
- Established reputation in integrative medicine practices
Consideration: When researching “Toxygen,” verify whether referring to Dotolo’s specific model or using term generically for closed-system equipment.
Emerging Asian Manufacturers Serving UK Market
The medical equipment landscape has evolved significantly. Asian manufacturers now produce CE-certified colon hydrotherapy machines meeting European regulatory standards while offering distinct value propositions for UK buyers.

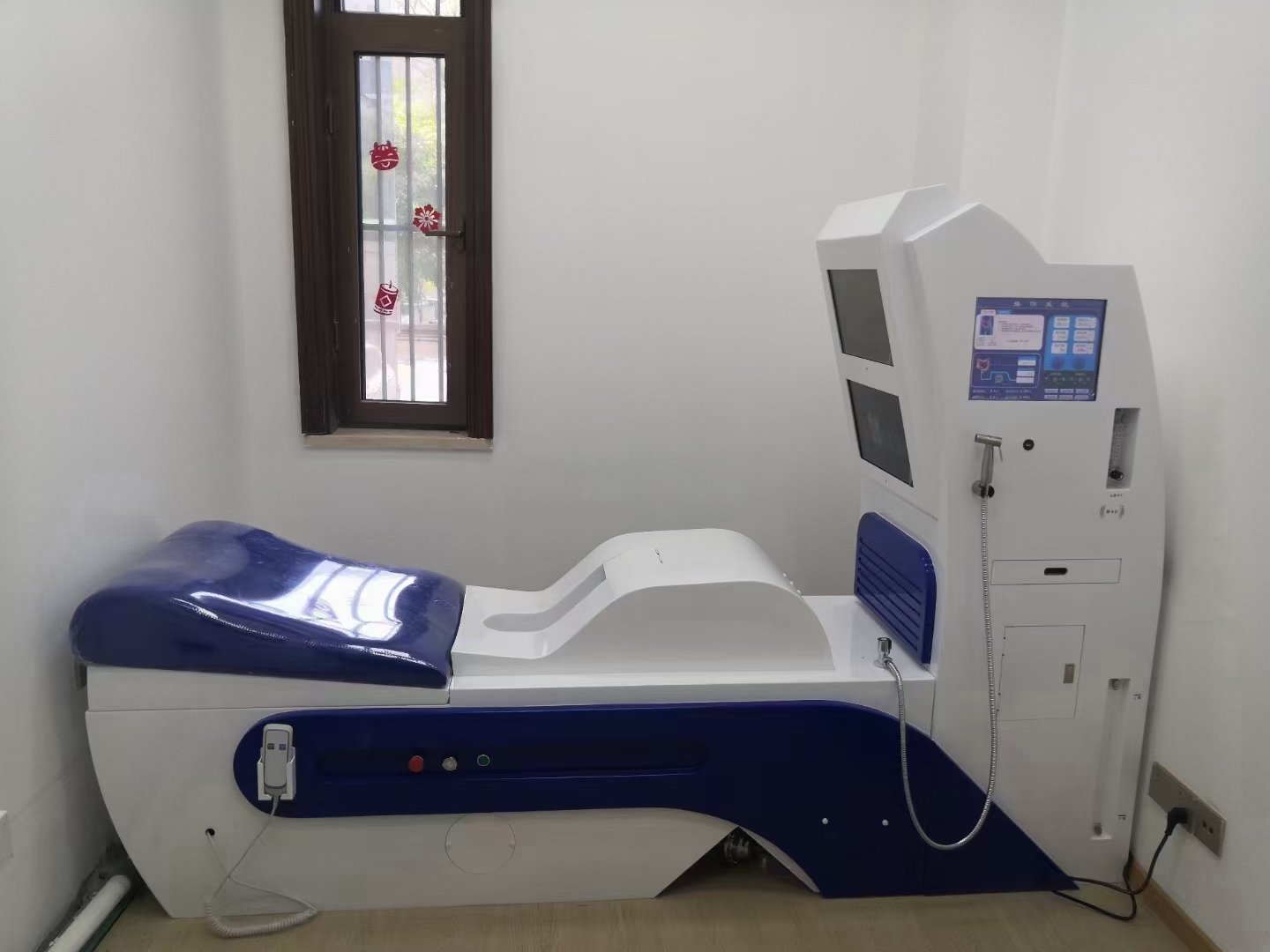
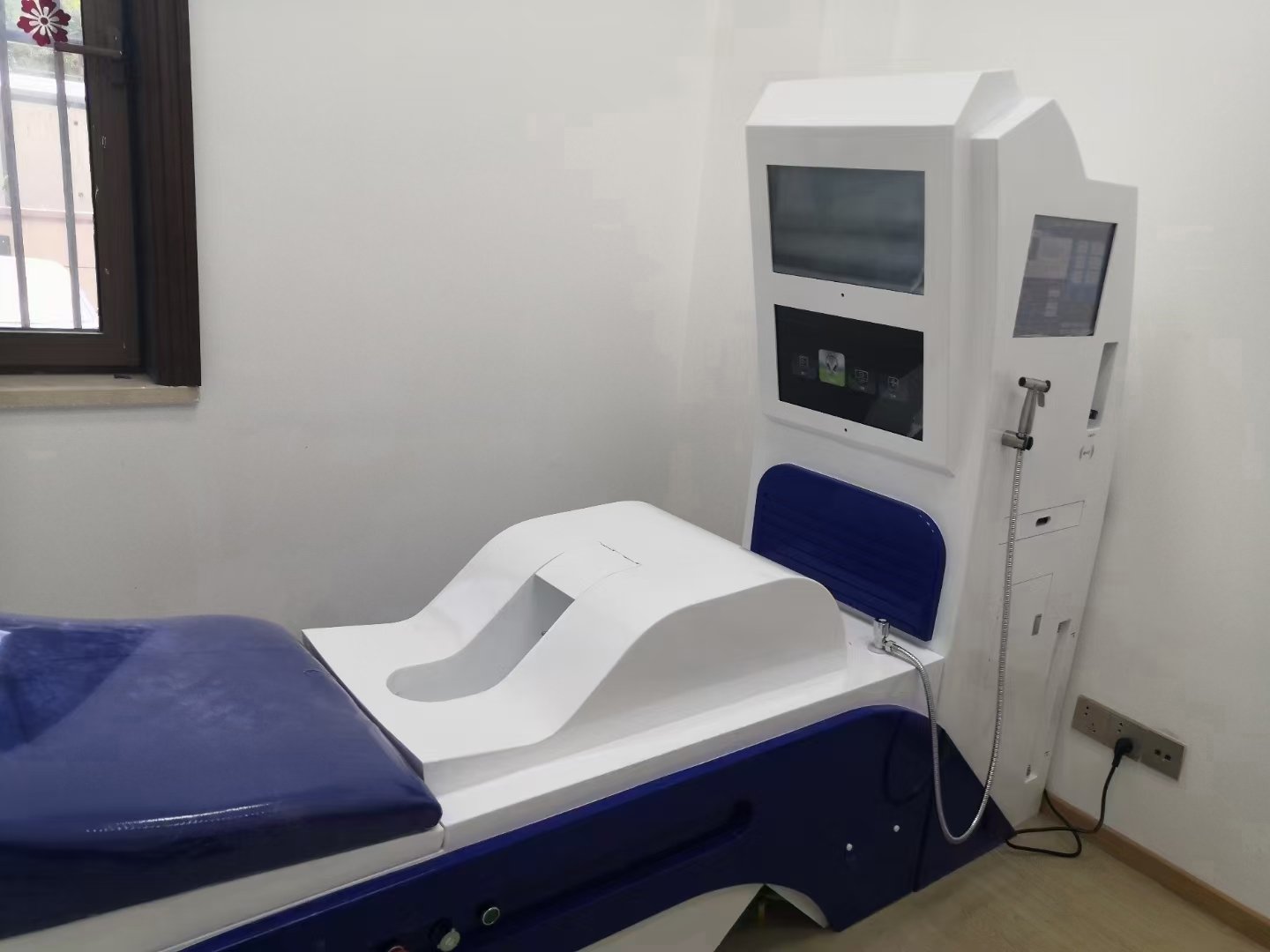
MAIKONG represents this emerging category, manufacturing in Shenzhen (GD, SZ) with advanced production facilities and quality control systems. I have examined their MK-880 model as a representative alternative:
MAIKONG MK-880 Technical Specifications
| Parameter | Specification |
| Model | MK-880 |
| Power Source | Electric – 220V-50HZ-2KW (UK compatible) |
| Dimensions | 1720mm × 2500mm × 690mm |
| Weight | 160kg |
| Water Tank Capacity | 10L |
| Medicine Storage Capacity | 500ML |
| Temperature Range | 30-40°C (adjustable) |
| Machine Noise Level | <65dB |
| Continuous Working Time | 8 hours |
| Quality Certification | CE certified for European market |
| Warranty | 1 Year standard |
| After-sale Service | Online technical support |
| Minimum Order Quantity | 1 Set (no bulk purchase requirement) |
| Delivery Timeline | 5-7 days |
| Packaging | Wooden case protective shipping |
MAIKONG MK-880 Package Contents and Accessories
| Component | Quantity | Purpose |
| Main Hydrotherapy Unit | 1 | Core treatment system |
| Water Filtration System | 1 set | Multi-stage water purification |
| Disposable Speculum Pack | 50 units | Initial consumables supply |
| Replacement Filter Cartridges | 3 sets | First-year filtration maintenance |
| Tubing Set (Disposable) | 50 sets | Single-use infection control |
| User Manual (English) | 1 | Operation and maintenance guidance |
| Installation Tools | 1 set | Setup and assembly |
| Training Video Materials | Digital access | Practitioner operation training |
The value proposition from manufacturers like MAIKONG centres on comparable technical specifications at significantly reduced pricing (typically 40-60% below European premium brands). For UK buyers prioritising cost-effectiveness while maintaining CE certification compliance, this represents a compelling alternative worth serious evaluation.
Reviewing colonic machines for sale across multiple suppliers provides market intelligence supporting informed procurement decisions.
UK Market Availability and Regulatory Compliance Considerations

Procuring colon hydrotherapy equipment for UK facilities requires navigating specific regulatory frameworks and understanding post-Brexit import dynamics. I have compiled essential compliance information UK buyers must address.
Medical Device Regulatory Requirements
The UK follows UKCA (UK Conformity Assessed) marking requirements for medical devices, though currently accepts CE marking during the transition period. Key compliance elements include:
- CE Marking Verification: Ensure equipment carries valid CE marking from notified body, indicating EU MDR 2017/745 compliance
- Declaration of Conformity: Request manufacturer’s DoC documenting regulatory compliance claims
- Technical Documentation: Obtain complete technical files demonstrating safety and performance validation
- UKCA Transition Planning: Understand timeline for potential UKCA marking requirements specific to your equipment category
- Registration Requirements: Register as a medical device user with MHRA if applicable to your facility type
When evaluating any colonic machine for UK deployment, verification of proper regulatory documentation protects your facility from compliance violations and ensures patient safety standards.
Import Considerations Post-Brexit
UK buyers face specific logistical considerations when importing from European or non-EU suppliers:
Direct Asian Import Advantages
- Single customs clearance process
- Potential duty savings versus intra-EU transfers
- Direct manufacturer relationship
- Faster shipping routes (air freight option)
- Transparent total landed cost calculation
European Distribution Considerations
- Additional distributor markup layers
- Complex EU-UK customs procedures
- Extended lead times through intermediaries
- Currency exchange fluctuations (EUR-GBP)
- Potential VAT complexity on intra-EU transfers
Dotolo’s European distribution model through Prague adds complexity UK buyers must evaluate against direct import alternatives. This structural consideration increasingly influences procurement decisions as buyers seek supply chain transparency and cost efficiency.
Warranty and After-Sales Support Infrastructure
Long-term equipment reliability depends significantly on accessible technical support and warranty service delivery. I have assessed typical support models:
Dotolo Support Structure
- European distributor manages warranty claims
- Technical support primarily via email/phone
- On-site service requires continental technician travel
- Spare parts shipped from European warehouse
- Extended response times for UK-based issues
Critical Consideration: Evaluate realistic service response times for your location. Remote facilities in Scotland or Northern Ireland may experience extended downtime if warranty service requires European technician deployment. Factor geographic service accessibility into your decision matrix.
Alternative suppliers offering UK-based support, parts inventory, or comprehensive remote troubleshooting capabilities may deliver superior total ownership experience despite potentially comparable warranty terms on paper.
UK Healthcare Facilities: Discuss equipment procurement, compliance verification, and partnership opportunities
WhatsApp available • English-speaking support • UK business hours accommodation
Professional Assessment: Dotolo Advantages and Limitations
After comprehensive evaluation, I have synthesised strengths and weaknesses UK buyers should weigh when considering Dotolo colonic irrigation machines for their facilities.
When Dotolo Represents Optimal Choice
Specific facility profiles and operational priorities align well with Dotolo’s value proposition:
- Established Medical Practices: Facilities with existing budgets for premium equipment and patients expecting recognised brand names
- Integrative Medicine Focus: Practitioners requiring ozone therapy integration alongside colon hydrotherapy capabilities
- High-Volume Clinics: Operations with sufficient patient throughput to justify premium investment through economies of scale
- Brand-Conscious Markets: Geographic markets or patient demographics showing strong preference for established North American/European brands
- Training Priority: Facilities valuing comprehensive practitioner education programmes included with equipment purchase
When Alternatives Merit Consideration
Conversely, certain operational contexts suggest exploring alternative equipment options:
- Start-up Clinics: New facilities managing capital constraints where cost-effective equipment preserves resources for marketing and operations
- Multi-Location Expansion: Groups purchasing multiple units where per-unit cost significantly impacts total investment
- Geographic Service Limitations: Remote UK locations where European-based support infrastructure creates operational risk
- Wellness-Focused Positioning: Facilities serving wellness rather than medical demographics may find open-system alternatives better suit client preferences
- Flexible Supplier Relationships: Operations prioritising direct manufacturer relationships, customisation options, or favourable payment terms
Your specific circumstances determine optimal equipment selection. No single brand universally suits all UK facilities. Honest assessment of your priorities, constraints, and patient demographic produces better long-term outcomes than brand reputation alone.
Practical Installation and Maintenance Considerations
Equipment procurement represents only the initial phase. Successful deployment requires addressing installation logistics and establishing sustainable maintenance protocols. I have outlined practical considerations UK facilities face.
Treatment Room Requirements and Setup
Proper installation ensures optimal equipment performance and regulatory compliance:
Physical Space Requirements
Adequate treatment room dimensions accommodate equipment footprint and patient movement:
- Minimum 3m × 3.5m room dimensions recommended
- Ceiling height 2.4m minimum for proper ventilation
- Dedicated electrical circuit (13A minimum capacity)
- Water supply connection with isolation valve
Environmental Controls
Climate management supports patient comfort and equipment longevity:
- Temperature control maintaining 20-24°C ambient range
- Adequate ventilation addressing odour management
- Appropriate humidity levels (40-60% relative humidity)
- Sound insulation ensuring patient privacy
Utility Infrastructure
Essential services must support equipment operational requirements:
- Water pressure 2-4 bar (typical UK mains pressure)
- Dedicated waste drainage with appropriate capacity
- Electrical supply meeting manufacturer specifications
- Emergency power-off accessible to practitioner
Infection Control Provisions
Hygiene protocols require specific room features:
- Washable wall and floor surfaces
- Hand-washing facilities within treatment room
- Appropriate storage for disposable supplies
- Waste management complying with clinical waste regulations
Maintenance Protocols and Schedules
Establishing systematic maintenance prevents equipment failures and ensures consistent treatment quality. I recommend UK facilities implement these protocols:
| Maintenance Task | Frequency | Responsible Party | Typical Duration |
| Pre-session System Check | Before each treatment | Practitioner | 5 minutes |
| Post-session Sanitisation | After each treatment | Practitioner | 10 minutes |
| Water Filter Replacement | Every 500 treatments or 6 months | Facility staff | 15 minutes |
| Deep System Cleaning | Monthly | Facility staff | 45 minutes |
| Calibration Verification | Quarterly | Qualified technician | 90 minutes |
| Comprehensive Service Inspection | Annually | Manufacturer/distributor | 2-3 hours |
Understanding comprehensive maintenance requirements helps you accurately forecast operational costs and staff time allocation. Equipment requiring complex maintenance procedures or expensive specialist servicing impacts total cost of ownership beyond initial purchase price.
For facilities evaluating best colonic machine options, maintenance accessibility and complexity represent critical selection criteria alongside clinical features and pricing.
MAIKONG: Strategic UK Market Partnership Opportunity


Having evaluated Dotolo comprehensively, I want to introduce you to an alternative worth your consideration. MAIKONG operates from Shenzhen, China (GD, SZ), manufacturing CE-certified colon hydrotherapy machines specifically designed for UK and European market requirements.
Why UK Facilities Choose MAIKONG
Our value proposition addresses the specific concerns UK healthcare buyers express when evaluating premium European brands:
MAIKONG Competitive Advantages
- 40-60% Cost Savings: Professional-grade equipment at significantly reduced investment versus European premium brands
- Direct Manufacturer Relationship: No distributor intermediaries – transparent pricing and direct technical support
- Flexible MOQ: Single-unit purchases available – no bulk order requirements
- Rapid Delivery: 5-7 day delivery timelines to UK locations
- CE Certification: Full European regulatory compliance documentation
- Customisation Options: Logo customisation and specification adaptations available
- UK Partnership Programme: Exclusive territory opportunities for qualified distributors
What MAIKONG Provides UK Partners
- Complete Technical Documentation: English-language manuals, installation guides, and training materials
- Online Technical Support: Responsive English-speaking engineering team addressing operational questions
- Marketing Materials: Professional product imagery, specification sheets, and promotional content
- Competitive Pricing Structure: Volume discounts and favourable payment terms for distributors
- Quality Assurance: Comprehensive testing protocols ensuring consistent manufacturing standards
- Ongoing Product Development: Continuous improvement incorporating market feedback and technological advances
MAIKONG MK-880: Technical Excellence Meets Economic Accessibility
Our flagship MK-880 model delivers clinical capabilities comparable to premium brands while maintaining accessible pricing for UK facilities:
- Advanced Water Filtration: Multi-stage purification removing chlorine, sediment, and contaminants
- Precise Temperature Control: Automatic heating maintaining physiological comfort zone (30-40°C adjustable)
- Quiet Operation: Sub-65dB noise levels ensuring patient relaxation and privacy
- Extended Duty Cycle: 8-hour continuous operation supporting high-volume clinic schedules
- Intuitive Controls: User-friendly interface minimising training requirements for practitioners
- Professional Construction: Medical-grade materials and components ensuring longevity and reliability
We understand UK buyers initially approach Chinese manufacturers with questions about quality and reliability. That scepticism serves you well – demanding evidence separates professional suppliers from questionable sources. I encourage you to request detailed specifications, certification documentation, and client references. Quality manufacturers welcome scrutiny.
Become a MAIKONG UK Distributor or Partner
We actively seek qualified partners to expand our UK presence. If you operate an established clinic, equipment supply business, or possess distribution capabilities within the UK healthcare sector, I invite you to explore partnership opportunities.
Ideal MAIKONG UK Partner Profile
What We Seek in Partners
- Established presence in UK healthcare, wellness, or medical equipment sectors
- Existing client relationships with private clinics, spas, or natural therapy practices
- Understanding of UK medical device regulations and compliance requirements
- Capability to provide local after-sales support and customer service
- Commitment to professional representation and quality standards
- Financial stability supporting inventory investment and business growth
What MAIKONG Provides Partners
- Exclusive territory options (where available)
- Competitive wholesale pricing structures
- Marketing and sales support materials
- Technical training and product knowledge resources
- Responsive manufacturer support and communication
- Flexibility in order quantities and specifications
Partnership Enquiry Process
Exploring partnership with MAIKONG involves straightforward steps respecting your time and business evaluation requirements:
- Initial Contact: Reach out via WhatsApp, email, or phone to introduce your business and express interest
- Information Exchange: We provide detailed product specifications, pricing structures, and partnership terms
- Sample Evaluation: Order a demonstration unit at preferential pricing to assess quality firsthand
- Terms Negotiation: Discuss territory, pricing, support arrangements, and contractual details
- Partnership Formalisation: Execute distributor agreement and establish operational procedures
- Ongoing Collaboration: Regular communication, market feedback, and business development support
Connect with MAIKONG UK Partnership Team
Whether you seek single equipment purchase for your facility or wish to explore distributorship opportunities across the UK market, our team provides responsive, professional support. We understand UK business practices and prioritise clear communication and reliable service delivery.
Visit our complete product range: colonicmachine.uk
Learn more about MAIKONG: About Our Company | Contact Information

Conclusion: Making Informed Colon Hydrotherapy Equipment Decisions
Dotolo colon hydrotherapy machines represent established technology with legitimate clinical applications and proven track record. For UK facilities where premium positioning, brand recognition, and specific feature requirements align with Dotolo’s offerings, the investment delivers value despite higher costs.
However, the medical equipment landscape offers diverse options. Your optimal choice depends on honest assessment of your facility’s priorities, patient demographics, budget constraints, and long-term operational goals. No shame exists in prioritising cost-effectiveness – redirecting capital toward marketing, staff development, or facility improvements often produces better business outcomes than premium equipment purchases.
I have presented comprehensive information about Dotolo equipment alongside credible alternatives. Your decision ultimately depends on factors unique to your situation. Whether you select Dotolo, competing European brands, or explore emerging manufacturers like MAIKONG, prioritise:
- Regulatory Compliance: Verify CE certification and complete documentation regardless of supplier
- Total Cost Analysis: Calculate five-year ownership costs including maintenance, consumables, and support
- Service Accessibility: Ensure realistic technical support and parts availability for your location
- Clinical Suitability: Match equipment capabilities to your therapeutic approach and patient needs
- Supplier Reliability: Assess manufacturer/distributor stability and communication responsiveness
The UK healthcare market benefits from competitive equipment options. Take advantage of that competition – request detailed quotations, compare specifications objectively, and negotiate favourable terms. Your informed purchasing decisions support both your business success and your patients’ wellbeing.
For those interested in exploring MAIKONG as a cost-effective alternative delivering professional capabilities, our team stands ready to provide detailed technical information, pricing transparency, and honest assessment of whether our equipment suits your specific requirements.
MAIKONG UK Partnership & Sales Enquiries:
Email: Lucy@colonicmachine.uk
English-speaking support team • CE-certified equipment • UK delivery available
Thank you for investing time in this comprehensive analysis. I wish you success in your equipment procurement process and the continued growth of your healthcare practice.
